 This protects the metal and stops it reacting. Unit C2: Further Chemical Reactions, Rates and Equilibrium, Calculations and Organic Chemistry. Magnesium reacts slowly when it is first added to water, but a layer of insoluble magnesium hydroxide forms. 2.1.1 recall the reactivity series of metals, including K, Na, Ca, Mg, Al, Zn, Fe and Cu 2.1.4 explain and describe the displacement reactions of metals with other metal ions in solution CCEA Double award science. 
The reactivity of a metal can be worked out by studying its reactions. Its surface naturally forms a very thin layer of aluminium oxide that keeps water away from the metal below. GCSE Edexcel Obtaining and using metals The reactivity series shows metals in order of reactivity. Aluminium is unusualīecause it is a reactive metal that does not react with water. In general, the more reactive the metal, the more rapid the reaction is. Sodium + water → sodium hydroxide + hydrogen For example, sodium reacts rapidly with cold water: periodic trends in properties of the elements, reactivity of elements. When a metal reacts with water, a metal hydroxide and hydrogen are formed. O Level Chemistry Notes PDF (IGCSE/GCSE Textbook): Class Notes Chapter 1-14 to. Reactivity series of metals Reaction of metals with water  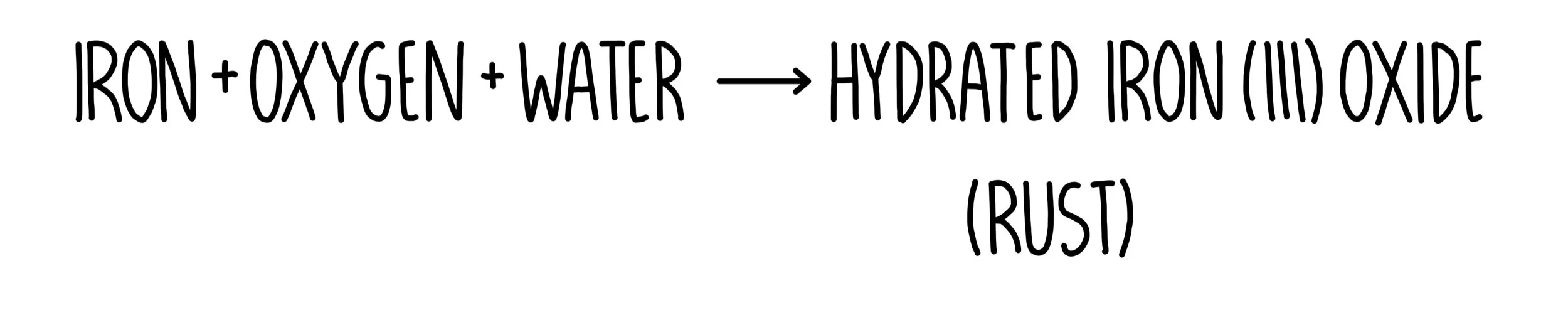
The table summarises some reactions of metals in the reactivity series. the more easily it loses electrons in reactions to form cations.In general, the more reactive a metal is: Provide rationale to explain why you chose these strategies.The reactivity series of metals is a chart showing metals in order of decreasing reactivity. InstructionsIn a 2-page paper, describe the care that Anna would require and address the questions below.What risk factors does Anna have that could predispose her to the development of cancer?What signs and symptoms could indicate that Anna has developed cancer?Based on Anna’s risk factors and presenting problems, identify three care strategies that you would use to provide quality care to Anna. AQA GCSE Chemistry 4: Chemical Changes Knowledge Organiser (Combined). Based on the physical findings Anna will undergo a diagnostic mammogram. KS3 Reactivity Series Differentiated Homework Worksheets. No abnormalities were found in the left breast. Anna’s right breast was tender upon palpation. No discharge from the nipple was observed. During the breast examination, the practitioner palpated a lump in Anna’s right breast. Anna has a positive family history of breast cancer both her mother and grandmother have been treated for breast cancer. She stated, “I am very busy with my children I haven’t had time to get it checked out.” She has recently been experiencing right nipple pain. During the history review, Anna shares with you that she has not been feeling like herself for the past six months, she has been unusually tired and thought that she felt a lump in her right breast during a self-breast exam around that same time. She a single mother of three teenagers and smokes regularly. Anna has a history of diabetes, obesity, and noncompliance with diet and medications to control her diabetes. ScenarioAnna is a 45-year-old female that presented to her physician’s office for her annual check-up. Module 01 ContentCompetencyExplain principles of care for clients with oncological disorders. This means that a more reactive metal will displace a less r. The reactivity of metals decreases going down the reactivity series. Some examples of metal-acid reactions and their equations are given below: When acids react with metals they form a salt and hydrogen gas: Sodium are very dangerous and react explosively with acids Metals that are placed high on the reactivity series such as potassium and The more reactive the metal then the more vigorous the reaction will be Only metals above hydrogen in the reactivity series will react with dilute acids This lesson bundle covers the content for the AQA GCSE Chemistry C5. Reaction with dilute sulfuric or hydrochloric acids 
The reactions with magnesium, iron and zinc and cold water are very slow The reactions of potassium and sodium have already been seen previously in theĪlkali metals, but the reaction with calcium and water is given here for reference:Ĭalcium + water ⟶ calcium hydroxide + hydrogen On the observations of their reactions with water and acids The series can be used to place a group of metals in order of reactivity based The chemistry of the metals is studied by analysing their reactions with water andīased on these reactions a reactivity series of metals can be produced 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
